1. Introduction
2. Materials and Methods
2.1. Identification and Phylogenetic Analysis of Lactic Acid Bacteria
2.2. Preparation of Whey
2.3. Fermentation of Strain and Whey
2.4. Measurement of pH and Titratable Acidity
2.5. Determination of Viable Cell Counts
2.6. Freeze-Drying of Fermented Whey
2.7. Storage Stability of Freeze-Dried Powder
3. Results and Discussion
3.1. Isolation and Identification of Lactic Acid Bacteria
3.2. Changes in pH and Titratable Acidity
3.3. Changes in Viable Cell Counts
3.4. Storage Stability of Freeze-Dried Postbiotic Powder
4. Conclusions
1. Introduction
Microbial fermentation has gained attention as a strategy to enhance the nutritional value of whey, improve its functional properties, and generate bioactive compounds. Among the microorganisms employed, lactic acid bacteria (LAB) are widely used due to their proven safety, technological advantages in fermentation, and recognized probiotic potential. Fermentation with LAB improves the digestibility of whey proteins through proteolysis and produces bioactive peptides with antioxidant, antimicrobial, and immunomodulatory properties [1]. Within this group, species of the genus Lactobacillus are particularly valued for their ability to survive gastrointestinal conditions, modulate the gut microbiota, and generate beneficial metabolites for the host. Lactobacillus plantarum is especially noteworthy due to its versatile metabolic adaptability, tolerance to acidic and bile environments, and strong proteolytic activity [2, 3]. Recent studies have reported that fructophilic lactic acid bacteria (FLAB) exhibit relatively low levels of antibiotic resistance, supporting their potential as probiotics or starter cultures. These characteristics are also relevant to the industrial application of honey-derived fermented products, providing essential baseline knowledge [4]. In recent years, postbiotics have emerged as a new paradigm in functional food research. During fermentation, microorganisms release bioactive compounds and enzymes that enhance the health-promoting value of the final product. Unlike probiotics, which require the presence and survival of viable cells within the gastrointestinal tract, postbiotics rely on the metabolites produced during fermentation and thus can exert biological activity regardless of cell viability [5]. Postbiotics comprise organic acids such as lactic and acetic acid, bacteriocins, bioactive peptides, exopolysaccharides, and cell wall components, which remain active even when cells are inactivated. For instance, L. plantarum and L. casei mainly produce lactic and acetic acids, while B. lactis additionally produces propionic and butyric acids. Supplementation with inulin has been shown to enhance the antioxidant activity of L. plantarum-derived postbiotics [6]. Moreover, postbiotics derived from L. plantarum have been reported to exert antimicrobial, antioxidant, and immunomodulatory effects [7]. These findings highlight postbiotics as safe, stable, and effective functional food components with promising applications in health promotion and disease prevention. The incorporation of prebiotic substrates further strengthens microbial fermentation by stimulating the growth and metabolic activity of probiotic strains.
Mongolian honey, harvested from wildflowers and medicinal plants in high-altitude regions, is characterized by its unique composition and superior bioactive properties compared to commercial honey. Natural honey is composed mainly of monosaccharides such as fructose and glucose, accounting for 70–80% of total carbohydrates, and also contains disaccharides, oligosaccharides, amino acids, vitamins, minerals, and polyphenolic compounds. In addition to its high proportion of monosaccharides (primarily fructose and glucose), honey typically contains 15–20% water, with moisture content serving as a key determinant of microbial stability and shelf life. The acidity of honey is characterized by a pH range of 3.2–4.5 and a free acidity of approximately 15–50 mEq/kg, conditions that contribute to its antimicrobial effect by inhibiting the growth of spoilage and pathogenic microorganisms. Honey also exhibits relatively high osmotic pressure due to its concentrated sugar matrix, further contributing to microbial inhibition [8, 9]. From a prebiotic perspective, honey contains oligosaccharides such as raffinose, kestose, and maltose, which can selectively stimulate the growth of beneficial lactic acid bacteria. Collectively, these physicochemical properties support honey’s role as an effective natural prebiotic substrate capable of enhancing microbial fermentation dynamics and promoting postbiotic metabolite production in dairy matrices. For the practical development of postbiotic-enriched fermented products, ensuring long-term stability while maintaining biological activity is a critical step. Lyophilization (freeze-drying) is considered a standard preservation technique for LAB and their metabolites, as it removes water under low temperature and vacuum conditions, thereby retaining microbial structural integrity and metabolite activity [10]. Converting fermented whey into stable postbiotic powders through freeze-drying provides wide applications in the functional food and nutraceutical sectors. Powder formulations offer advantages such as extended shelf life, ease of transportation, and controlled application across delivery systems, while maintaining consistent bioactivity without being dependent on microbial viability [11].
Therefore, the present study aims to: (i) determine the impact of honey supplementation on the fermentation behavior and metabolic activity of L. plantarum in whey; and (ii) evaluate the feasibility and physicochemical stability of converting the resulting fermented matrices into postbiotic powders through freeze-drying. We hypothesize that honey supplementation will enhance microbial growth and metabolism, promote the formation of bioactive postbiotic metabolites, and ultimately produce freeze-dried powders with superior antioxidant capacity, functional stability, and technological suitability compared to non-supplemented controls.
In conclusion, integrating honey supplementation with whey fermentation and subsequent postbiotic powder production represents a promising approach consistent with current global priorities in sustainable food processing, valorization of dairy by-products, and circular bioeconomy practices. The outcomes of this study are expected to support the development of high-value, shelf-stable postbiotic ingredients derived from whey and honey, offering potential applications across functional foods, nutraceutical formulations, and health-oriented product development.
2. Materials and Methods
2.1. Identification and Phylogenetic Analysis of Lactic Acid Bacteria
Mongolian artichoke (Cynara scolymus L.) was subjected to anaerobic fermentation at 37°C for 30 days, during which lactic acid bacteria were isolated. For bacterial isolation, De Man Rogosa-Sharpe (MRS, Difco, USA) agar was employed, supplemented with 0.02% sodium azide (Sigma, St. Louis, MO, USA) for selective growth. After fermentation, colonies differing in size, morphology, and color were selected and purified on MRS agar. The metabolic capacity for carbohydrate utilization of the selected strains was assessed using the API 50 CHL kit (BioMerieux Co., Marcy-I’Etoile, France). A single colony was inoculated into sterilized CHL medium, dispensed at 100 µL per capsule, and incubated at 37°C for 48 h, after which color changes were examined. The resulting fermentation patterns were analyzed using API web software (https://apiweb.biomerieux.com/) to determine preliminary identification. For final identification, 16S rDNA sequencing of the isolates was outsourced to a professional analysis institution (Solgent, Korea). The obtained sequences were compared with those of reference strains registered in international databases, and final identification was based on the strain showing the highest sequence homology.
2.2. Preparation of Whey
Whey used in the experiment was produced from commercially available whole milk through enzymatic coagulation. Whole milk was transferred into a sterilized double-jacket stainless-steel container and heated to 35 ± 1°C to maximize enzymatic activity. Once the target temperature was reached, rennet (Danisco, France) was added at a final concentration of 0.02% (w/v). The mixture was gently stirred for 1–2 min to ensure uniform enzyme distribution and then left undisturbed at 35°C for 30–40 min to allow coagulation. Upon completion of coagulation, curd and whey were distinctly separated. The whey fraction was carefully collected and filtered through sterilized muslin cloth to remove residual curd, yielding a clear supernatant. The pH of the filtrated whey was measured and adjusted to 7.0 using 1 N NaOH solution. Finally, to ensure microbiological safety and suitability for subsequent fermentation experiments, the neutralized whey was sterilized by autoclaving at 125°C for 20 min.
2.3. Fermentation of Strain and Whey
Whey, a representative by-product of the dairy industry, was fermented using Lactobacillus plantarum isolated from artichoke (Cynara scolymus L.) to enhance its functional value. Prior to inoculation, the isolated strain was subcultured twice consecutively in De Man, Rogosa, Sharpe (MRS) broth (Difco, USA) to ensure consistent metabolic activity and growth stability. Fermentation experiments were carried out with two treatment groups. (1) The control group consisted of sterilized whey inoculated with 2% (v/v) of actively cultured L. plantarum. (2) The combination group consisted of sterilized whey inoculated with 1% (v/v) of L. plantarum together with 1% (w/v) of natural honey (Selenge, Mongolia) to evaluate the potential prebiotic effect of honey. For each treatment, 300 mL of sterilized whey was dispensed into sterile Erlenmeyer flasks and inoculated according to the specified conditions. Fermentation was conducted under static conditions at 37°C for 24 h. The pH of the fermentation broth was measured at 3 h intervals, and the endpoint of fermentation was defined as the time when pH reached approximately 4.5.
2.4. Measurement of pH and Titratable Acidity
During fermentation, pH and titratable acidity (TA) were measured every 3 h over a 24 h period. The pH was directly measured using a pH meter (Mettler-Toledo, USA). Titratable acidity was determined following the standard method of the Association of Official Agricultural Chemists (AOAC). Specifically, 17.6 mL of fermented whey was diluted with distilled water at a 1:1 ratio, and then titrated with 0.1 N NaOH solution until pH 8.3 was reached. The final acidity value was calculated according to the following equation:
2.5. Determination of Viable Cell Counts
The viable cell counts during fermentation were measured at 12 h intervals. Fermented whey and honey-supplemented whey samples were homogenized, and 1 mL of each sample was serially diluted with 9 mL of sterile 0.1% (w/v) peptone water. Subsequently, 1 mL of each dilution was spread onto MRS agar (Difco, USA). Plates were incubated at 37°C for 24–48 h, after which colonies were enumerated. Results were expressed as the average colony-forming units (CFU) per mL of sample, calculated from triplicate measurements.
2.6. Freeze-Drying of Fermented Whey
To improve the storage stability and functional integrity of fermented whey, lyophilization was performed under aseptic conditions. Fermentation was carried out at 37°C for 24 h, and immediately after completion, whey samples were cooled at 4°C to suppress microbial metabolic activity. Prior to freeze-drying, samples were pre-frozen at –80°C for at least 12 h to ensure the formation of uniform and optimized ice crystals. The frozen samples were then dried using a laboratory-scale freeze-dryer (IlShinBioBase, Korea). The drying process was conducted under high-vacuum conditions, with the condenser temperature maintained at –50°C and the chamber pressure below 0.1 mbar. The drying cycle lasted approximately 48 h, and complete dehydration was considered achieved when the weight of the samples became stable. The resulting freeze-dried fermented whey powders were immediately sealed in sterilized polypropylene tubes and stored at 10°C. This process enabled the long-term preservation of probiotic and postbiotic components, which were subsequently used for stability and functional evaluations.
2.7. Storage Stability of Freeze-Dried Powder
The storage stability of freeze-dried postbiotic powders was assessed by monitoring changes in viable cell counts over a storage period of six months. For analysis, 1 g of each powder sample was suspended in 9 mL of sterile 0.1% (v/v) peptone water and serially diluted using a 10-fold dilution method. From each dilution, 1 mL was spread onto MRS agar (Difco, USA). Plates were incubated at 37°C for 24–48 h, after which colony numbers were enumerated. Final results were expressed as colony-forming units (CFU) per mL of sample, and values were calculated as the mean from triplicate measurements.
3. Results and Discussion
3.1. Isolation and Identification of Lactic Acid Bacteria
Artichoke (Cynara scolymus L.) was fermented under anaerobic conditions for 30 days, after which the samples were homogenized and streaked onto MRS agar supplemented with sodium azide. The cultures were incubated anaerobically at 37°C until colonies were formed. Selected colonies were analyzed for carbohydrate utilization using the API 50 CHL kit, and the results indicated 99.9% homology with Lactobacillus plantarum. Furthermore, 16S rDNA sequencing and homology searches against established databases also revealed approximately 99% identity with the same species, thereby confirming the isolates as L. plantarum.
3.2. Changes in pH and Titratable Acidity
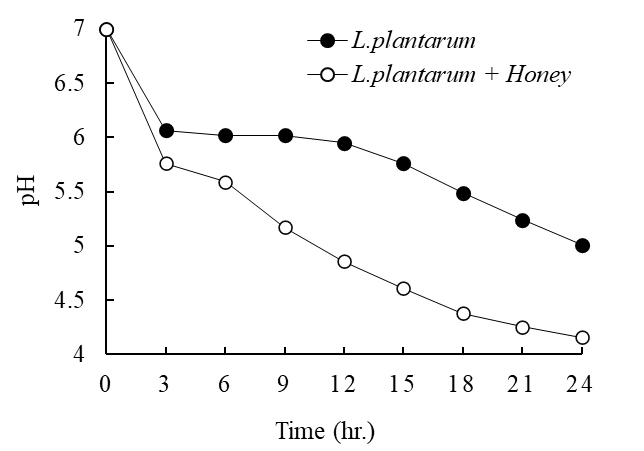
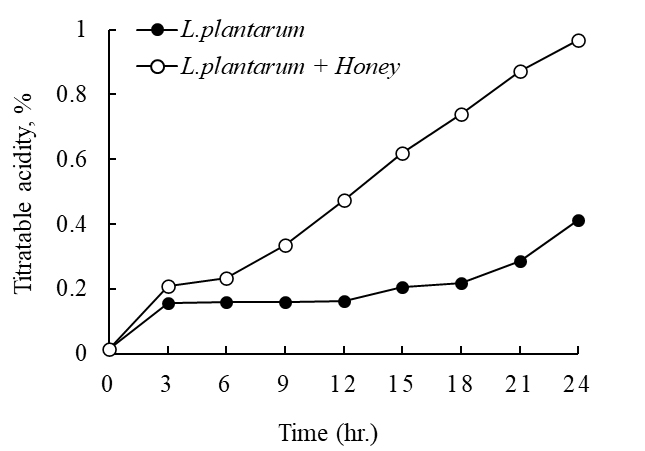
Distinct differences in pH and titratable acidity were observed between the L. plantarum single inoculation group and the honey-supplemented fermentation group during 24 h of fermentation. In the control group inoculated with L. plantarum alone, the pH decreased sharply within the first 3 h of fermentation, but the rate of decrease slowed between 6 h and 15 h, showing a gradual change thereafter. By the end of fermentation at 24 h, the pH had reached approximately 5.0. In contrast, the honey-supplemented group exhibited more vigorous acidification from the early stages of fermentation, with the final pH stabilizing at approximately 4.16 (Fig. 1). This trend suggests that the monosaccharides, oligosaccharides, polyphenols, and various nutrients contained in honey promoted the growth and metabolism of L. plantarum, thereby accelerating the accumulation of organic acids, particularly lactic acid. Titratable acidity also differed between the two groups. In the control group, acidity gradually increased during fermentation, reaching approximately 0.41% after 24 h. In comparison, the honey-supplemented group showed a more pronounced and steady increase, with final acidity reaching approximately 0.96% at 24 h (Fig. 2). These results indicate that the carbohydrates and bioactive components of honey strongly stimulated the fermentative metabolism of L. plantarum, significantly enhancing both the rate and level of acid production compared with the control group. Taken together, these findings support the role of honey as an effective prebiotic substrate during L. plantarum fermentation. This suggests that whey fermented with honey supplementation could be developed into a functional dairy product with stable acidity and improved probiotic activity.
3.3. Changes in Viable Cell Counts
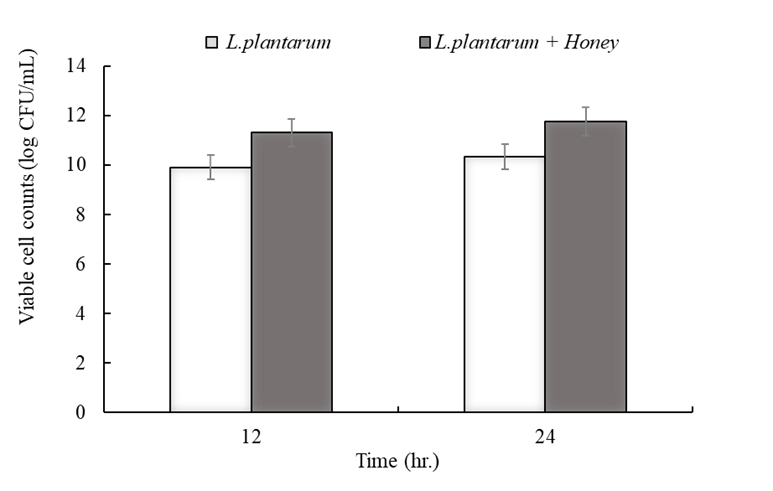
The changes in viable cell counts of L. plantarum during 24 h of whey fermentation at 37°C are shown in Fig. 3. In the control group fermented with L. plantarum alone, the number of viable cells gradually increased with time, reaching approximately 10.3 log CFU/mL at the end of fermentation. In contrast, the honey-supplemented group exhibited significantly higher viable counts, reaching approximately 11.9 log CFU/mL. This increase can be attributed to the immediate availability of simple sugars, such as glucose and fructose, in honey, which served as direct energy sources to stimulate glycolytic activity. Honey is composed of sucrose, glucose, fructose, maltose, and other sugars, with oligosaccharides accounting for about 10% of its total weight [12]. Along with these carbohydrates, the amino acids and trace minerals in honey act as prebiotic substrates that promote microbial growth and metabolism. Research from 2025 showed that milk fermented with 5% polyfloral honey maintained the highest probiotic viability at 72.41% with 1.26 × 10⁶ CFU/ml, with honey's sugars serving as energy sources for microbial metabolism [13]. Furthermore, phenolic compounds and other bioactive constituents present in honey suppress the proliferation of competing microorganisms, thereby providing a selective environment favorable for the dominance of L. plantarum during fermentation [12]. Thus, the higher viable counts observed in the honey-supplemented fermentation group can be explained as the result of these multiple synergistic effects. These findings highlight the potential of honey as an effective prebiotic substrate in whey fermentation, providing an important basis for the development of functional fermented dairy products with enhanced probiotic content.
3.4. Storage Stability of Freeze-Dried Postbiotic Powder
The changes in viable cell counts during six months of storage of whey fermented with L. plantarum for 24 h and subsequently freeze-dried are presented in Fig. 4. At one month of storage, both groups maintained viable counts at approximately 11–12 log CFU/mL; however, the counts gradually declined over time, reaching about 6 log CFU/mL after six months. This decrease reflects reduced viability due to structural cell damage and nutrient depletion during long-term storage. Although the honey-supplemented group exhibited higher viable counts during the fermentation stage, its long-term stability after freeze-drying was somewhat lower than that of the control group. During the freeze-drying process, rapid freezing can cause ice crystal formation, which may damage the cell membrane, while the drying stage leads to removal of water molecules, resulting in changes in the structural arrangement of proteins and lipids and increasing the likelihood of denaturation. Consequently, some cells are inactivated; however, osmoprotectant compounds in honey, such as trehalose, sugars, and polyphenols, contributed to maintaining partial cell survival. In fact, viable counts remained at levels of 8–9 log CFU/mL during 3–4 months of storage, and even after six months, the counts were still above the acceptable threshold for functional dairy products. On the other hand, the high sugar content and hygroscopicity of honey promoted moisture absorption during storage, lowering the glass transition temperature (Tg) and leading to stickiness and aggregation. Additionally, glucose crystallization further accelerated powder hardening and clumping. The present results demonstrate that freeze-drying enables the preservation of L. plantarum and its metabolites for a certain period; however, to improve long-term stability, the application of additional protective strategies such as cryoprotectant supplementation or encapsulation is required. Therefore, this study suggests that while honey contributes positively to fermentation and short-term storage stability, supplementary approaches are necessary to achieve industrial-scale production of stable postbiotic powders.
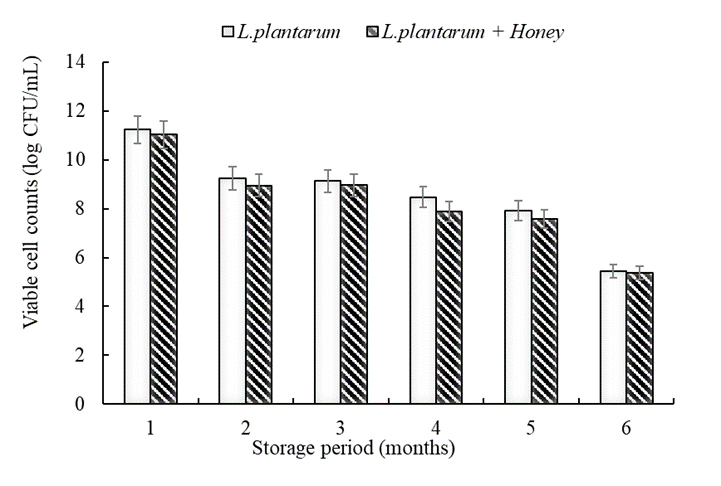
Fig. 4
Changes in the number of viable cells during storage. Whey fermentation was performed at 37℃ for 24 hours, then freeze-dried and stored at room temperature for 6 months. During this time, viable cell counts were determined monthly by serial dilutions in peptone water, followed by a 48-hour incubation at 37℃. The number of colonies formed was counted and expressed as.
4. Conclusions
In this study, whey fermentation was conducted using Lactobacillus plantarum, and the effects of honey supplementation as well as the storage stability after freeze-drying were comprehensively evaluated. During fermentation, L. plantarum induced a marked decrease in pH and an increase in titratable acidity within 24 h, with viable cell counts ultimately reaching approximately 11–12 log CFU/mL. In particular, the honey-supplemented group exhibited synergistic effects on microbial growth and metabolic activity, demonstrating that honey can function as an effective prebiotic substrate. After freeze-drying and storage at room temperature for six months, high viable counts were maintained initially but gradually decreased over time, reaching approximately 6 log CFU/mL by the end of the storage period. Nevertheless, this level still satisfied the minimum standard for probiotic products (≥6 log CFU/mL). Although the honey-supplemented group showed relatively higher viable counts immediately after fermentation, its long-term stability was slightly lower than that of the control group. These results suggest that cell membrane damage, protein denaturation, and oxidative stress during freezing and storage were the primary causes of viability loss, while certain protective factors present in honey, such as trehalose, sugars, and polyphenols, contributed to partial survival. Therefore, this study demonstrates that honey supplementation can enhance probiotic activity and physiological functionality during whey fermentation and that freeze-drying enables the preservation of viable cells and postbiotic components at a certain level over long-term storage. However, to further improve long-term stability, additional technological approaches such as cryoprotectant supplementation or encapsulation are required. Collectively, the findings indicate the potential of honey-based fermented whey for enhancing functionality and for the development of stable probiotic and postbiotic ingredients.